|
4/10/2024 0 Comments Absolute entropy of ccl4
Let us look at charging a battery, and creating new surface by distorting a spherical drop of liquid. Let us now look at two entirely different situations, both involving non- PdV work. Many of the examples of thermodynamical calculations have hitherto involved PdV work in a system in which the working substance has been an ideal gas. The CCl4 is nonpolar in nature because of the symmetrical tetrahedral geometrical structure. This slight puzzle will remain with us until Chapter 16, when we meet Nernst’s Heat Theorem and the Third Law of Thermodynamics. The entropy appearing in equations 12.9.9 and 12.9.11 is surely the absolute entropy, and we cannot calculate this unless we know the entropy at T = 0 K. After all, all we have done in this example is to calculate the increase in entropy as we took the sample up to 25 oC and 1 atmosphere – we haven’t really calculated the absolute entropy. But this leaves us in a rather uncomfortable position. Now that we have calculated the absolute entropy at a given temperature and pressure, we can calculate the increase in the Helmholtz and Gibbs functions from equations 12.9.9 and 12.9.11. Hence, taking the entropy to be zero at 0 K, the required entropy is 124000 J K −1 kmole −1. 2 STATE FUNCTIONS A property of a system which depends only on its present state and not on its pathway. There is no molecular motion at absolute 0 K. The third law of thermodynamics The entropy of a perfect crystal at 0 K is zero. See equation 12.9.4, from which we see that there is a decrease of entropy equal to R ln( P 2/ P 1) = 8314ln(1.103 × 10 5 / 7173) = 22000 J K −1 kmole −1. is always an increase in the entropy of the universe. Increase pressure to 1 atmos = 1.013 × 105 Pa at constant temperature. Assuming that we know C P as a function of temperature in this range, that comes to 70000 J K −1 kmole −1.ĥ. The increase in entropy is ∫ C P d(ln T). Increase temperature to 298.15 K at constant pressure. Study with Quizlet and memorize flashcards containing terms like Calculate the standard-state entropy of reaction for the following reactions and explain the sign of S° for the below reaction Hg(l) Hg(g) Hg(l) S° 76.02 J/mol·K Hg(s) S° 174. The molar latent heat of vaporization is 911000 kmole −1. Vaporize it at the same temperature and pressure. (b) Calculate the enthalpy of vaporization of methanol, assuming that rS doesnt depend on temperature and taking the boiling point of methanol to be 64.6C. (a) Calculate the entropy change for the vaporization of 1 mol methanol (use data from Table 16.1 or Appendix J). Increase in entropy = 117000/13.95 = 8400 J K −1 kmole −1.ģ. The standard molar entropy of methanol vapor, CH3OH(g), is 239.8 J K1 mol-1. The molar latent heat of fusion is 117000 J kmole −1. Liquefy it at the same temperature and pressure. change in enthalpy (H), change in entropy (S), and absolute temperature. Assuming that we know C P as a function of temperature in this range, that comes to 2080 J K −1 kmole −1.Ģ. enthalpy of the carbonoxygen bond in carbon monoxide A CO(g) C(s) + O(g). (That’s the triple point.) The increase in entropy is ∫ C P d(ln T). Heat the solid hydrogen from 0 K to 13.95 K at a pressure of 7173 Pa. 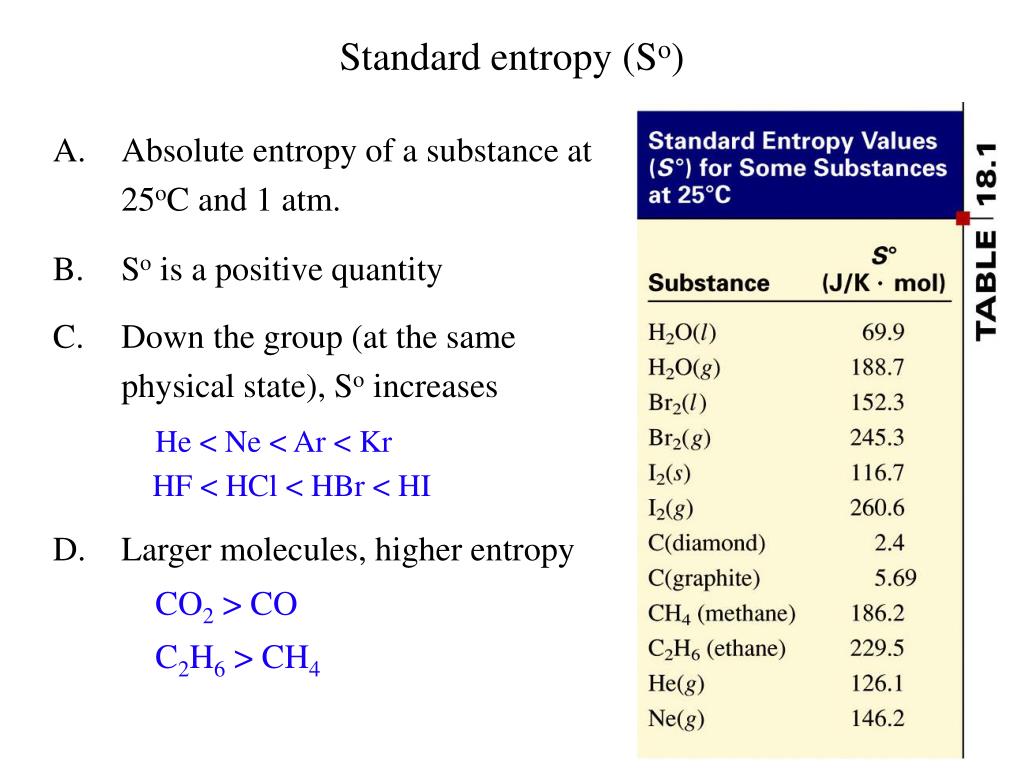
You will find it helpful to sketch these stages on a drawing similar to figure VI.5.ġ. 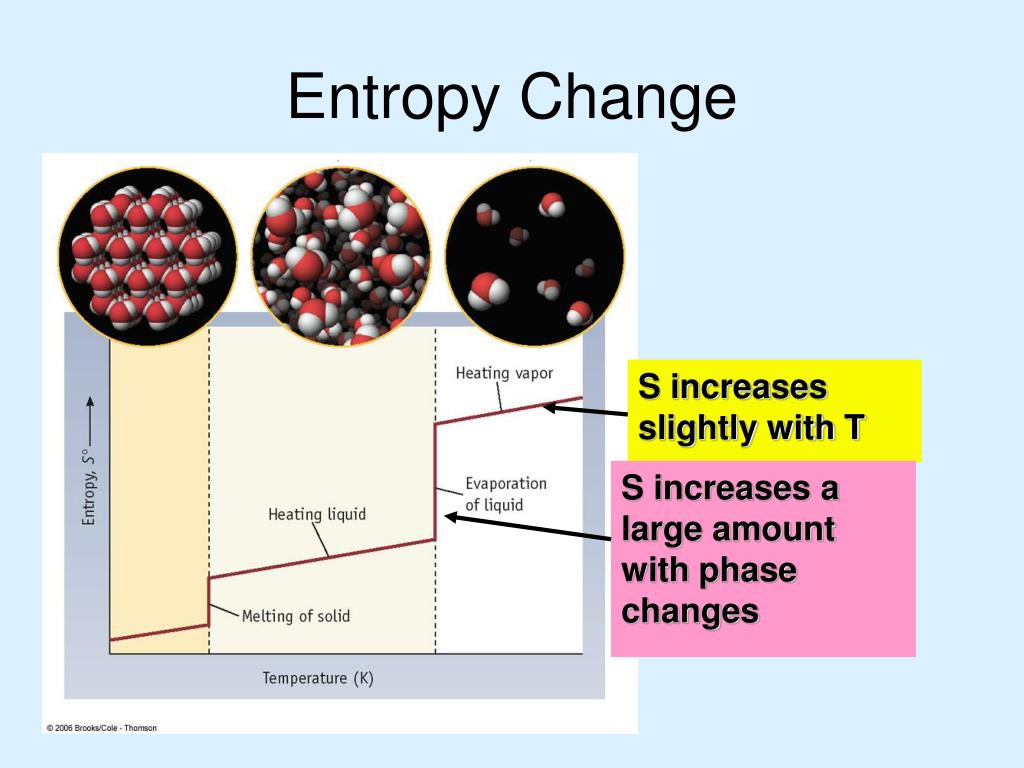
1.00 mol of carbon disulfide vapour condenses to liquid at 25 degree C. Related to this Question The heat of vaporization of carbon disulfide, CS2, at 25 degree C is 27.2 kJ / mol. We can do this in five stages, as follows. Explore the definition of absolute entropy and how the third law of thermodynamics applies to absolute entropy in this lesson. By way of example, assuming that the molar entropy of hydrogen at 0 K is zero, calculate the absolute entropy of a kmole of H2 gas at a temperature of 25 oC (298.15 K) and a pressure of one atmosphere. We can, of course, calculate the molar entropy of a substance at some temperature provided that we define the entropy at a temperature of absolute zero to be zero. The reactions are listed in the following table.\) A student was asked to propose three reactions for the oxidation of carbon or a carbon compound to CO or CO 2. (In Chapter 13, we see that the entropy changes for the mixing and separation processes are important.) When we call (DeltarHo) the standard enthalpy change for the reaction, we are indulging in a degree of poetic license.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
